


Invion is partnering with world leading research institutes to develop a novel photosensitiser, based on the Photosoft technology, that may become a viable alternative to treating cancers and infectious diseases. Photosoft aims to be more effective at targeting and killing cancers and other insidious diseases without harmful side effects and minimal discomfort.
Invion produced a new topical and intravenous product suitable for use in skin cancer, infectious diseases and solid tumours, such as those present in ovarian, prostate and lung cancers.
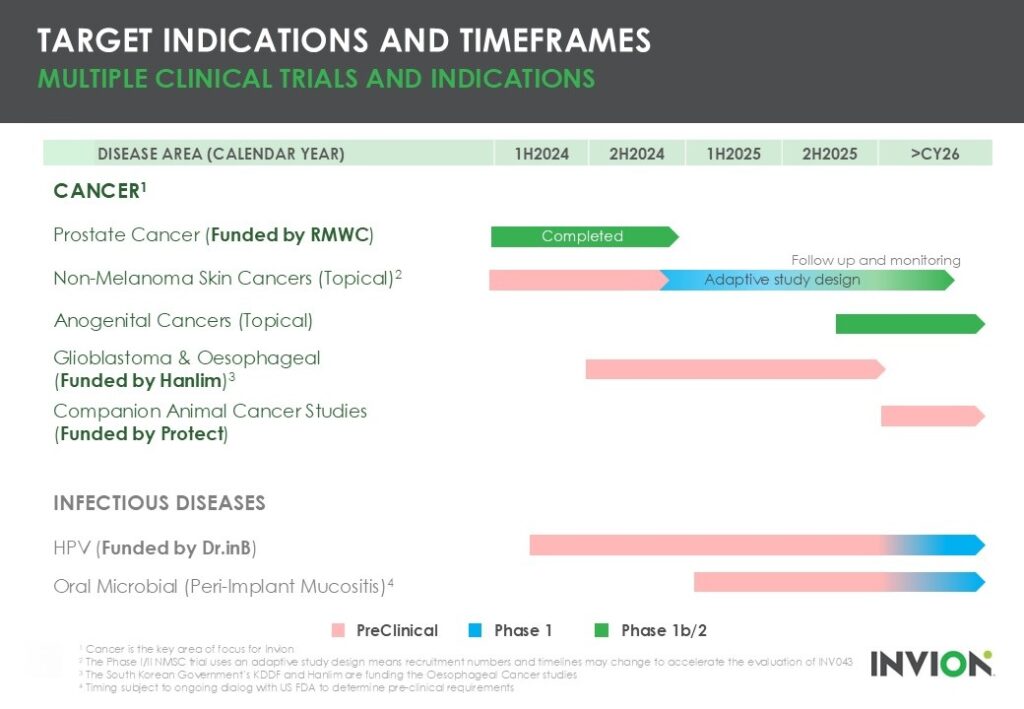
Given the significant commercial and clinical potential of its assets, Invion will look to progress multiple programs and commence pre-clinical and clinical trials in collaboration with leading global cancer research institutes such as the Hudson Institute of Medical Research and Peter MacCallum Cancer Centre.
The Company’s priority is in cancers and it has published results from a Phase II prostate cancer trial and is undertaking a Phase I/II non-melanoma skin cancer (NMSC) study in Queensland.
Invion will also further explore the application of its technology in anogential and lung cancers.
Find out more about Invion’s areas of interest by clicking on the buttons below.
